The experts at Bent Glass Design are here today to set the record straight. And contrary to what you may have heard in the past, it’s not as complicated as you might think.
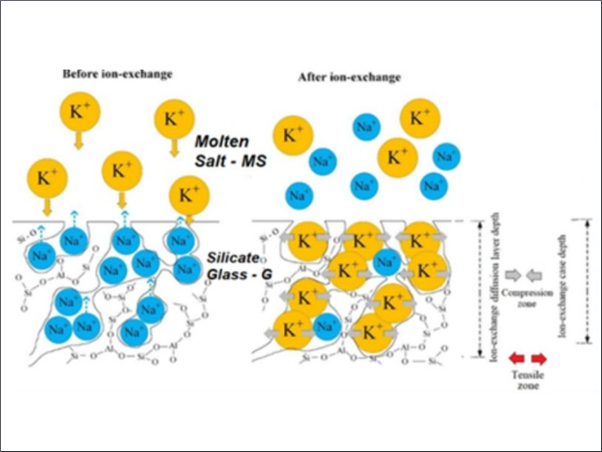
Chemically strengthened glass is glass that has been treated with a chemical process after it was manufactured to render it stronger. The chemical process in question is called an ion-exchange reaction.
During this process, glass is submerged in a bath of potassium salt or potassium nitrate at 300 degrees Celsius. The bath isn’t hot enough to melt the glass, but the relatively high temperature allows the potassium nitrate to react with the surface of the glass, causing ions to be exchanged and compacted, thereby strengthening the glass.
The glass sits in the bath for up to 30 hours in order for the chemical reaction to be complete, and then it is removed. The end result is called chemically strengthened glass.
Glass that has been chemically strengthened is usually about six to eight times the strength of float glass, which is another type of strengthened glass made from floating melted glass on molten metal, like tin.
This is the kind of glass you’d see on a modern building facade. Float glass is typically uniformly thick and smooth. It’s strong, but chemically strengthened glass is stronger.
Chemically treated glass will break differently than regular glass. Rather than shattering into many small pieces, it will break into larger shards that are not as sharp as untreated glass. Chemically treated glass is also far less prone to warping and distorting over time than other types of glass.
For more information on chemically treated glass, or to learn more about any of the other types of glass we work with here at Bent Glass Design, contact us today, or check out our gallery.