This paper was first presented at GPD 2025.
Link to the full GPD 2025 conference book: GPD_2025_ConferenceProceedingsBook.pdf
Authors: Guglielmo Macrelli a, Elisabetta Poli b, Airy Cortada c, Alex Viera c
a. Isoclima SpA R&D Department, SiMathModels, Italy
b. Isoclima SpA R&D Dept., Italy
c. Quality Chemicals, Spain
Abstract
A mathematical model is presented for the concentration of incoming ions in the glass and the residual stress build-up resulting from the ion exchange processes between silicate glasses and a bath of molten alkali nitrate salts. The role of quality characteristics of molten salt is discussed. The discussion covers both thermodynamic equilibrium issues at glass surface and interface between glass and the molten salts and the interdiffusion non-equilibrium kinetics. Measurement methods and results are reported for surface compression (SC) and compression layer depth (Cd) for different soda-lime glasses: clear, low-iron and grey body tinted. The discussion of the salt chemistry related to ion exchange chemical strengthening is focused on the service life of plants in the industrial environments. Salts pollution ad their de-activation mechanisms are introduced and critically discussed. Conclusions are summarized and presented putting in evidence the importance and relevance of this study to the application areas of silicate glasses chemical strengthening by ion exchange.
Article Information
- Published by Glass Performance Days, on behalf of the author(s)
- Published as part of the Glass Performance Days Conference Proceedings, June 2025
- Editors: Jan Belis, Christian Louter & Marko Mökkönen
- This work is licensed under a Creative Commons Attribution 4.0 International (CC BY 4.0) license.
- Copyright © 2025 with the author(s)
1. Introduction
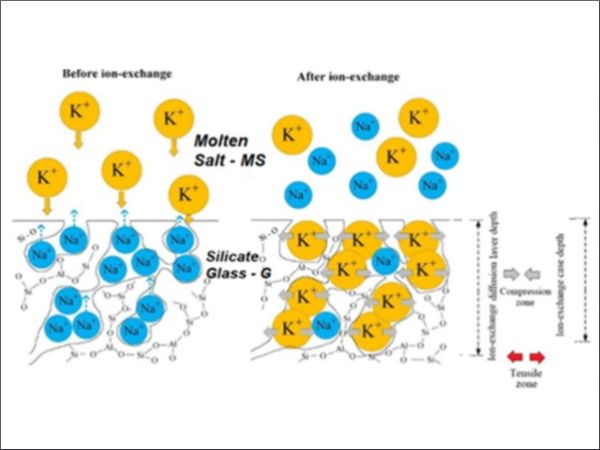
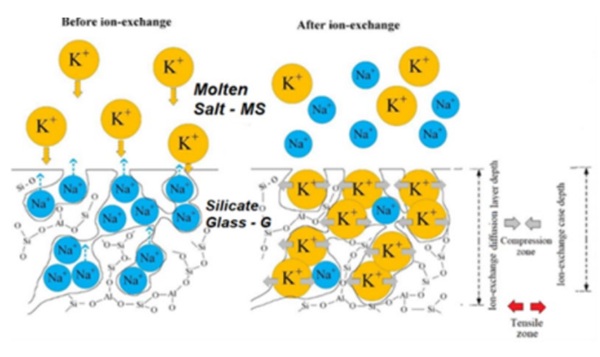
Silicate glasses usually present strength values in the order of few tens of MPa with a significant statistical dispersion. In structural applications it can be requested a higher strength and reduced dispersion of strength values. This can be accomplished by increasing glass strength through technological strengthening processes like thermal tempering or chemical strengthening. In this study we focus on chemical strengthening of silicate glasses by ion exchange processes. The scientific background of these processes is known since several decades (Gy (2008), Karlsson et al (2010) even though some residual issues are still debated in the scientific community (Varshneya (2010); Macrelli, Varshneya and Mauro(2019)). We refer here to the classical process of ion exchange where glass articles are immersed in a bath of molten nitrate salts. In these circumstances alkali ions (usually Sodium) in the glass matrix are exchanged for other alkali ions (usually Potassium) present in the molten salts. Based on this statement, ion exchange is possible only if alkali or other exchangeable ions are present in the glass matrix. For this reason we will focus on Alkali Silicate Glasses which, nevertheless, represent the wider class of glass materials used in industrial applications. Chemical strengthening by Ion-Exchange is presently used in applications where it is used thin glass (t<2mm) which cannot be practically strengthened by thermal tempering or in glass articles with a complex surface geometry (again not viable for thermal tempering) or for glass articles that shall retain a high level of surface quality after strengthening processes. The role of the molten salts in the chemical strengthening process and its contaminations has been extensively discussed in the scientific literature (Zhang, He, Xu and Zheng (1986), Sglavo (2015), Talimian and Sglavo (2017), Sglavo Talimian and Ockso (2017), Hassani and Sglavo (2019)). In these past studies, the effects of the contamination of the molten salt bath due to Sodium, divalent ion species like Calcium, Magnesium Strontium and Barium and anionic groups are considered and discussed. It has been found that contamination at parts per million concentration levels may significantly affect the ion exchange process. In the present study our purpose is to discuss the role of Alkaly Nitrate Salts from two perspectives: limitation in terms of quantity of molten salt versus glass volume or glass surface to effectively drive the ion exchange process efficiently and effect of contaminations that can influence strengthening performances. In Figure 1 it is depicted the Ion Exchange process between a glass and a molten salt where Sodium in the glass matrix is exchanged for Potassium in the molten salt.
The equilibrium condition for ion exchange is represented by the following equation:
![]()
where the subscripts “g” and “MS” indicate the glass matrix and the molten salts medium respectively. From the equilibrium condition (1) it is evident that the first contaminant of the molten salts bath is the Sodium itself. The thermodynamic equilibrium conditions have been extensively discussed by Macrelli (2024a) in terms of chemical potentials of the ions and it has been evaluated the effect of the increase of concentration of Sodium in the molten bath to the surface concentration of Potassium in the glass. Recently Kirchner and Mauro (2023) have identified in the entropy production in the molten salt and in the glass, the driving force of ion exchange and, specifically, they calculated that the major fraction of entropy production is in the salt bath which represents more than 90% of total entropy produced in ion exchange. One of the main factors to maximize entropy production in the salt bath is the excess of salt bath to glass volume ratio. As demonstrated by Macrelli (2024b), the excess of salt bath volume together with the absence of limitations to the diffusion of Potassium ions in the salt bath are necessary conditions to keep the kinetics of ion exchange at optimized level. Chemical Strengthening by ion exchange can be understood as a thermodynamic and kinetics problem leading to an equilibrium thermodynamic condition at glass surface and a kinetic process of ions interdiffusion in the glass bulk.
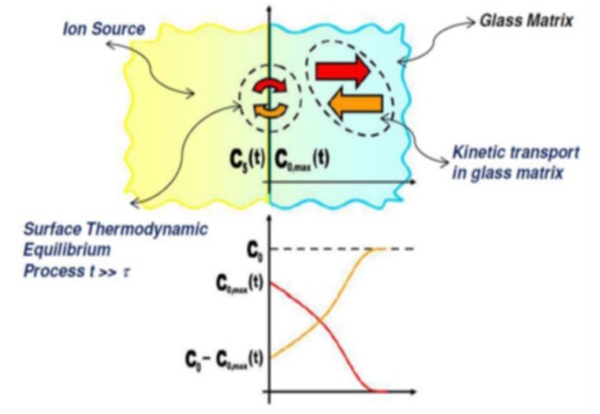
The surface thermodynamic condition makes sense if equilibrium at glass surface is achieved in a time (τ) much lower than the overall immersion time of glass in the salt bath (t). This assumption allows to define the interdiffusion kinetic problem with a boundary condition of constant equilibrium surface concentration (Cs). In other words, we assume that surface concentration of incoming ions is achieved as glass is immersed in the molten salt bath and maintained at a fixed level for the total immersion time. This assumption translates in mathematical terms the above introduced conditions of excess of available ions in the molten salts and the absence of any obstacle to their availability for ion exchange. When the two exchanging ions have different volumes and, namely, the incoming ion is larger than the outgoing one, the consequence is a constrained expansion of the near surface layer (the exchanged surface layer tends to expand while the inner not exchanged part of glass body constraints this expansion) leading to the build-up of a residual stress in the glass, characterized by a compression state in the near surface volume balanced by a tensile state in the glass bulk. The compressive stress layer works to inhibit crack formation and propagation increasing glass strength and resistance to damages. As outlined above and clearly expressed by Gy (2008), glass strength is not an intrinsic property, reflecting the statistical distribution of the severity – i.e. of the depth – of surface flaws. The evaluation of the strengthening effect due to ion exchange needs a fracture mechanics approach where the interaction of the introduced residual stress field with the characteristics of surface flaws is considered. This last issue is not the subject of the present study.
2. Mathematical Model of Ion exchange and experimental methods.
2.1. Mathematical model of stress build-up in glass by ion exchange
The chemical reaction indicated in equation (1) indicates that the ion exchange is an equimolar reaction where the same number of moles, N (mol), of Sodium and Potassium are exchanged between glass and molten salt. Because the molar weight of the exchanging ions is different, the first obvious consequence is that ion exchange generates a weight gain ∆W of glass
![]()
where MW is the molecular weight (g/mol) of the exchanging ions. The size (Volume) difference of the exchanging ions generates a strain condition in the near surface glass layer occupied by the incoming ions, if the temperature at which ion exchange occurs is far below the glass transition temperature, the near surface strain results uncompensated and translates into a residual stress characterized by compression in the near surface glass layer balanced by tensile state in the glass bulk. This residual stress condition is the cause of the glass strength increase. The strengthening effect resulting from ion exchange in glass is related to the concentration of the incoming ions in glass (mol/cm3) that can be expressed, assuming a mono-dimensional case, by C(x,t) that is a function of spatial coordinate x and time t.. The stress build-up is due to the introduction of the larger incoming ions into the sites previously occupied by the smaller ones which generates an uncompensated strain. The stress profile along glass thickness σ(x,t) can be derived (Tandia et al.(2012)) from the concentration profile of the incoming alkali ions C(x,t). Taking the assumption of negligeable relaxation effects the stress profile after ion exchange can be expressed according to equation (3):
![]()
where E and v are respectively the Young modulus and Poisson ratio of the glass, Cavg is the average of C(x,t) along glass article thickness. B is the linear network dilation coefficient, in other words it represents the linear strain per unit change in alkali concentration. From equation (3) it can be derived surface compression Sc:
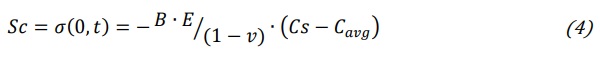
where Cs is the concentration of incoming ions at the glass surface, Cs=C(0,t). The condition for the determination of the compression layer depth Cd is simply in correspondence to the zero value of stress profile:
![]()
The condition (5) according to equation (3) results:
![]()
Hence, Cd is the value of x where stress profile changes from compression to tensile which, in turn, according to equation (3), is the point where the concentration profile is equal to its average value. It is worth to remember that negative values of stress represent compression while positive values represent tensile stress. The coordinate x where there is the transition from compression of the near surface layer to the tensile status of the inner portion of glass is just the value of the compression layer depth which is mathematically represented by equations (5) and (6). Based on this model it is evident the central role of the concentration of the incoming ions. All equations from (3) to (6) can be conveniently used providing that we have an expression for the incoming ions concentration in the glass. The crucial point to come to a concentration profile expression is the boundary condition at glass/molten salt interface. If we have an excess of available ions in the bath (volume of molten salts Vb >> of glass volume Vg) and no limitation conditions to ions transfer from the bath to glass we can assume that, as glass is immersed, surface concentration Cs is almost instantaneously achieved and so maintained for the whole immersion time. Under this boundary condition, and assuming a constant interdiffusion coefficient D, the concentration profile can be obtained as solution of the diffusion equation and it is expressed through the complementary error function (erfc) (Varshneya and Mauro (2019) :
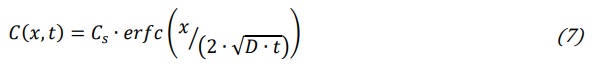
where D is the interdiffusion coefficient representing the effective diffusion coefficient of the incoming ions. The average concentration can be calculated from equation (7) as follows:
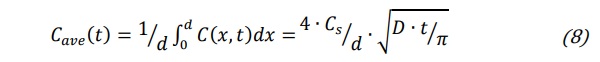
Where d is the glass article thickness.This simple mathematical model allows the prediction of the depth of compression layer. This approach can also be considered for the determination of the interdiffusion coefficient. Assuming that the diffusion of the incoming ions follow the first Fick law the flux of incoming ion is:

Based on equation (9) the total exchanged molar flux, FKNa is
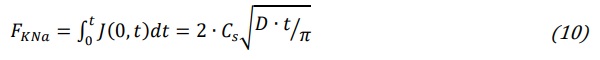
The exchanged molar flux has dimensions (mol/cm2). Following equation (1) and assuming an equimolar exchange, the number of exchanged moles of alkali ions (N) can be obtained from equation (2) dividing the weight increase ∆W of a glass sample after ion exchange by the difference of molar weight of the two exchanged ions (MwK-MwNa). Defining the exchange surface S which is the total glass surface exposed to ion exchange, the molar flux (mol/cm2) can be measured by measuring the weight increase ∆W of a glass sample:
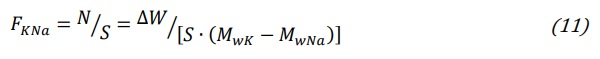
From equations (10) and (11) the interdiffusion coefficient is:

The value of surface concentration (Cs) of Potassium can be, at maximum, the value of initial concentration of Sodium. The exchange factor (γ) represents the ratio of surface concentration and original Sodium concentration (CNa):

It results that the exchange factor is limited between 0 and 1: 0 ≤ γ ≤ 1. Typically, and for efficient Ion exchange processes it is expected that at minimum γ=0.9.
2.2. Experimental Part: characterization of molten salt bath by ion exchange test
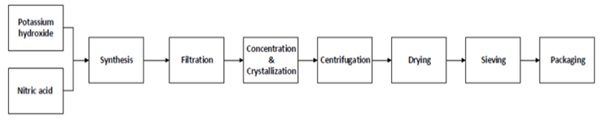
Potassium Nitrate is an inorganic salt available at different levels of purity. We have to distinguish salt coming from mining extraction and further purified and salt coming from chemical synthesis of Potassium Hydroxide and Nitric Acid:

As already discussed even small contaminants of Calcium, Magnesium and Sodium may significantly reduce the ion exchange performances of the salt. It is quite evident that purity level and contamination control can be performed at any requested level by the chemical synthesis process as described in Figure 3, The control of contaminants in the Potassium Nitrate is primarily achieved by the control of the purity of the precursors of reaction (14) and, at a second level, by the control of all production steps indicated in the production scheme of Figure 3.
The salt coming from mining extraction, even though purified as indicated in figure 4, may contain uncontrolled contaminants that may interfere in a negative way with the ion exchange of glass.
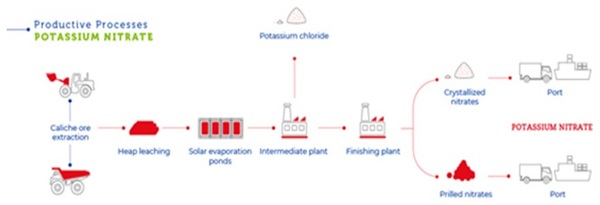
The presence of anionic groups like sulfates and chlorates together with an excess of [OH]- groups may drive to a chemical corrosion attack to glass surface. The above arguments suggest that the best way to characterize Nitrate Salts for ion exchange applications is to perform an ion exchange functional test using a small quantity of salt and glass samples. The Nitrate Salt selection based only on a chemical analysis is not sufficient to guarantee an efficient ion-exchange process. Usually in a chemical analysis it is found the presence of what you look for, that means there can be a contamination level at a part per million concentration that can negatively influence the ion exchange process. In this study we have used a quantity of 6kg of Potassium Nitrate provided by Quality Chemical, manufactured by chemical synthesis and identified with the code:145804 with a typical chemical analysis reported in Table 1, this salt is identified for our purposes: KNO3-Synt. An additional, test was performed in a different tank also with a technical grade (Assay 99.8%) Potassium Nitrate (KNO3-Prilled) coming from mineral extraction, further purified and prepared in prilled form (see Figure 4, where “prilled” means that anticaking agents is introduced to avoid agglomeration of salt).
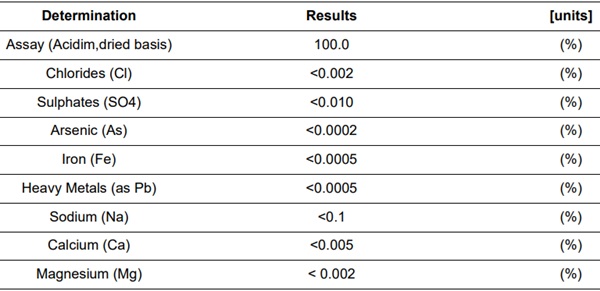
Table 1: Typical analysis of KNO3 – Qemical ID:145804 (KNO3-Synt).
Four rectangular samples of soda-lime glass, namely clear (CLP) thickness 1.6mm and (CL) float, thickness 6mm, low iron (EX) float, thickness 6mm and grey (GE) float thickness 6mm have been submitted to ion exchange in a test performed at 450°C with an immersion time of 24h.The glass samples with nominal dimensions 66x66mm have been cleaned and weighted on an analytical balance with a resolution of 0.1 mg. The samples have been introduced in a supporting stainless steel (AISI 316 L) basket than loaded in the pre-heating chamber of the test device and further immersed in the bath. The pre-heating time is such that samples are heated at not less than 420 °C before immersion in a tank of stainless steel (AISI 321) containing the molten salt. After the 24 hours immersion samples have been extracted with no post heating. The cooling down to room temperature is performed to allow washing out of residual salts on the surface of the samples. After careful drying samples have been optically inspected to check any possible surface chemical attack or corrosion. After inspection samples have been weighted again with the same analytical balance to determine weight increase after ion exchange. Surface compression Sc (MPa) and compression layer depth Cd (µm) has been measured on all samples using a FSM 6000 differential surface refractometer manufactured by Orihara (Japan).The instrument has a uncertainty of +/- 3µm for case depth Cd and +/-30MPa for surface compression. The total volume of glass samples in each ion exchange test is Vg=83.4 cm3 with a total exchange surface of Sg=400.2 cm2, while the volume of salt bath is Vb=3000 cm3. The relevant parameters are the volume ratio Vg/Vb and the exchangeable surface to volume ratio Sg/Vb (cm⁻¹).

After this comparison tests, additional tests were performed at four different temperatures (440°C,450°C,460°C,470°C), using the KNO3-Synt (Qemical ID: 145804) and using soda-lime float glass (CLP) to determine the dependence on ion exchange temperature of the interdiffusion coefficient D(T).
3. Results and Discussion
3.1. Molten salt comparison
A comparison based on ion exchange characteristics for a Potassium Nitrate salt manufactured by chemical synthesis (KNO3-Synt) and a Potassium Nitrate salt manufactured by mineral extraction a purified and finally prilled (KNO3-Prilled) is presented in table 2.
Table 2: Strengthening characteristics [Sc(MPa and Cd(µm)] and chemical attack (Corr) on the different sodalime glasses for the two different salts.
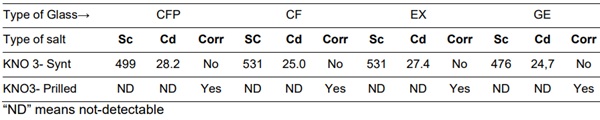
Results reported in Table 2 clearly indicate that the KNO3-Prilled salt type is not suitable for ion exchange and namely chemical strengthening of glass. We suspect that the main adverse issue is related to the chemical agent used to avoid agglomeration of salt crystallite to get the “prilled” almost spherical shape.
3.2. Determination of temperature dependence of the interdiffusion coefficient
Knowing the interdiffusion coefficient as a function of molten salt temperature D=D(T) and knowing the immersion time it is possible to calculate the concentration profile of the incoming ions and, consequently, it is possible to evaluate the compression layer depth Cd. As outlined by Gy (2008), interdiffusion coefficient has an Arrhenius type behaviour with temperature according to the following expression:

where Ea (kJ) is the activation energy for the hopping diffusion of ions from one site to another.
This activation energy depends on the glass chemical composition and structure. R is the gas constant and T is the absolute glass temperature in Kelvin at which diffusion occurs while D0 is a pre-exponential factor. The determination of the D(T) can be performed by measuring the values of D for a number of temperature points following the method outlined in section 2.1 using equation (12). This approach has been considered in the present study for glass type CFP which has the chemical composition reported in table 3.
Table 3: Chemical composition of soda-lime glass CFP used for process control (Main Components).

Ion exchange tests has been performed at four immersion temperatures 440°C, 450°C, 460°C and 470°C. The interdiffusion coefficient has been determined by weight gain and evaluated according to equation (12). Results are reported in Table 4.
Table 4: Interdiffusion coefficient D(cm2/s) for the ion exchange K+⇄Na+ for soda-lime glass CFP.

In Figure 6 a plot is reported with the experimental point and the regression curve which allow the determination of the pre-exponential factor D0 and of the activation energy Ea:
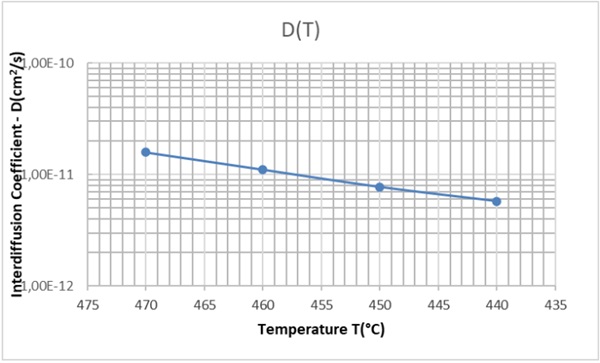
From the best fit procedure applied to the experimental results (Table 4) and represented by the continuous line of figure 6 it results:

The activation energy (17)b is in a good agreement with the value reported by Gy (2008) for soda-lime glass.
4. Conclusions
From the results presented it can be concluded that the proposed method can be conveniently considered for the determination of the parameters of chemical strengthening by ion exchange. The limitation of the method is that it can be applied for ion exchange processes where stress relaxation can be considered negligeable that mean within certain limitation of temperatures and immersion times. The method can also be used for prediction of strengthening parameters for different molten salt temperatures and immersion times. The application of the evaluation method to different types of molten salts of Potassium Nitrate puts in evidence that, the presence of contaminants can be so detrimental that no glass strengthening is detectable and, moreover, surface chemical attack may develop corroding glass surface. This conclusion means that a great attention shall be focussed about the selection of molten salt quality. A qualification protocol can be established based on the methodology proposed in this study. Another important element to be considered is the need to have an excess of salt volume in respect to glass volume and exchangeable surface. In this study we have used a volume ratio higher than a factor of 30. Making sure of an excess of molten salt volume versus glass volume or exchangeable surface guarantees that surface concentration of incoming ions can be considered constant and not influenced by the enrichment of Sodium of the bath as a result of ionexchange. Surface concentration of incoming ions is strictly correlated with surface compression, hence constancy of surface concentration implies in some way also constancy of surface compression. A method has been introduced to evaluate interdiffusion coefficient as a function of temperature based on an Arrhenius function. This allows to predict compression layer depth for ion exchange processes for different temperatures and immersion times.
References
Gy, R (2008),Ion exchange for glass strengthening, Materials Science and Engineering B 149, 159-165.
Hassani, H and Sglavo, V. (2019), Effect of Na contamination on the chemical strengthening of soda-lime silicate float glass by ion exchange in molten potassium nitrate. Journal of Non-Crystalline Solids, 515, 143-148.
Karlsson,S, Jonson B, Stalhandske C,(2010), The technology of chemical glass strengthening – A review, Glass technology: European journal of glass science and technology part A, 51(2), 41-54.
Kirchner, K and Mauro, J.C. (2023), Entropy production during ion exchange of glass, J Amer Ceram Soc., 107, 880-896, https://doi.org/10.1111/jace.19534.
Macrelli, G., Varshneya, A.K. and Mauro, J.C.(2019), Simulation of glass network evolution during chemical strengthening: Resolution of the subsurface compression maximum anomaly, Journal of non-crystalline solids Volume 522, 15 October 2019, 119457, https://doi.org/10.1016/j.jnoncrysol.2019.05.033.
Macrelli,G. (2024a), Thermodynamics and kinetics of silicate glasses submitted to ion exchange: equilibrium conditions and interdiffusion kinetics, https://doi.org/10.48550/arXiv.2401.10916.
Macrelli, G. (2024b), Diffusion in a two phases system: application to ion exchange in silicate glass, https://doi.org/10.48550/arXiv.2406.10818
Talimian, A. and Sglavo V.M. (2017), Ion exchange strengthening of borosilicate glass: influence of salt bath impurities and treatment temperatures, Journal of Non-Crystallyne Solids, 456: 12-21.
Tandia, A, Vargheese, K.D., Mauro, J.C., Varshneya, A.K. (2012),Atomistic understanding of network dilation anomaly in ion exchanged glass., Journal of Non Crystalline. Solids, 358, 316-320.
Varshneya, A.K. (2010), Chemical strengthening of glass: lessons learned and yet to be learned, International journal of applied glass science, https://doi.org/10.1111/j.2041-1294.2010.00010.x.
Varshneya, A.K. and Mauro, J.C.(2019), Fundamentals of inorganic glasess 3rd Edition, Amsterdam: Elsevier.
Sglavo, V.M. (2015), Chemical strengthening of soda-lime silicate float glass effect of small differences in KNO3 bath : International Journal of Applied Glass Science, 6, 72-82. DOI: https://doi.org/10.1111/ijag.12101.
Sglavo, V., Talimian, A. and Ockso (2017), Influence of salt bath calcium contamination on soda lime silicate glass chemical strengthening, Journal of Non-Crystalline Solids, 458, 121-128.
Zhang, X, He, O, Xu, C, Zheng, Y (1986), The effect of impurity ions in molten salt KNO3 on ion-exchange and strengthening of glass; Journal of Non-Crystalline Solids, 80, 313-318.